The Purse Group at San Diego State University is an organic chemistry research team focused on the molecular design, synthesis, and mechanistic study of functional DNA and RNA.
We use the tools of synthetic and physical organic chemistry to create artificial chemical modifications of nucleic acids and to study how molecular structure controls photophysics, reactivity, and function in complex nucleic acid environments. A central theme of our work is establishing structure–property relationships that enable rational molecular design.
Our research integrates organic synthesis, spectroscopy, physical organic analysis, and biochemistry to develop minimally perturbing chemical tools for investigating nucleic acid structure, dynamics, and interactions, and artificially modifying their functions in chemical biology and biotechnology.
Research Highlights
Purse Lab paper: Chem Sci 2025, 16, 4866–4875.
Single-molecule detection of oligonucleotides using the fluorescent nucleobase analogue ABN
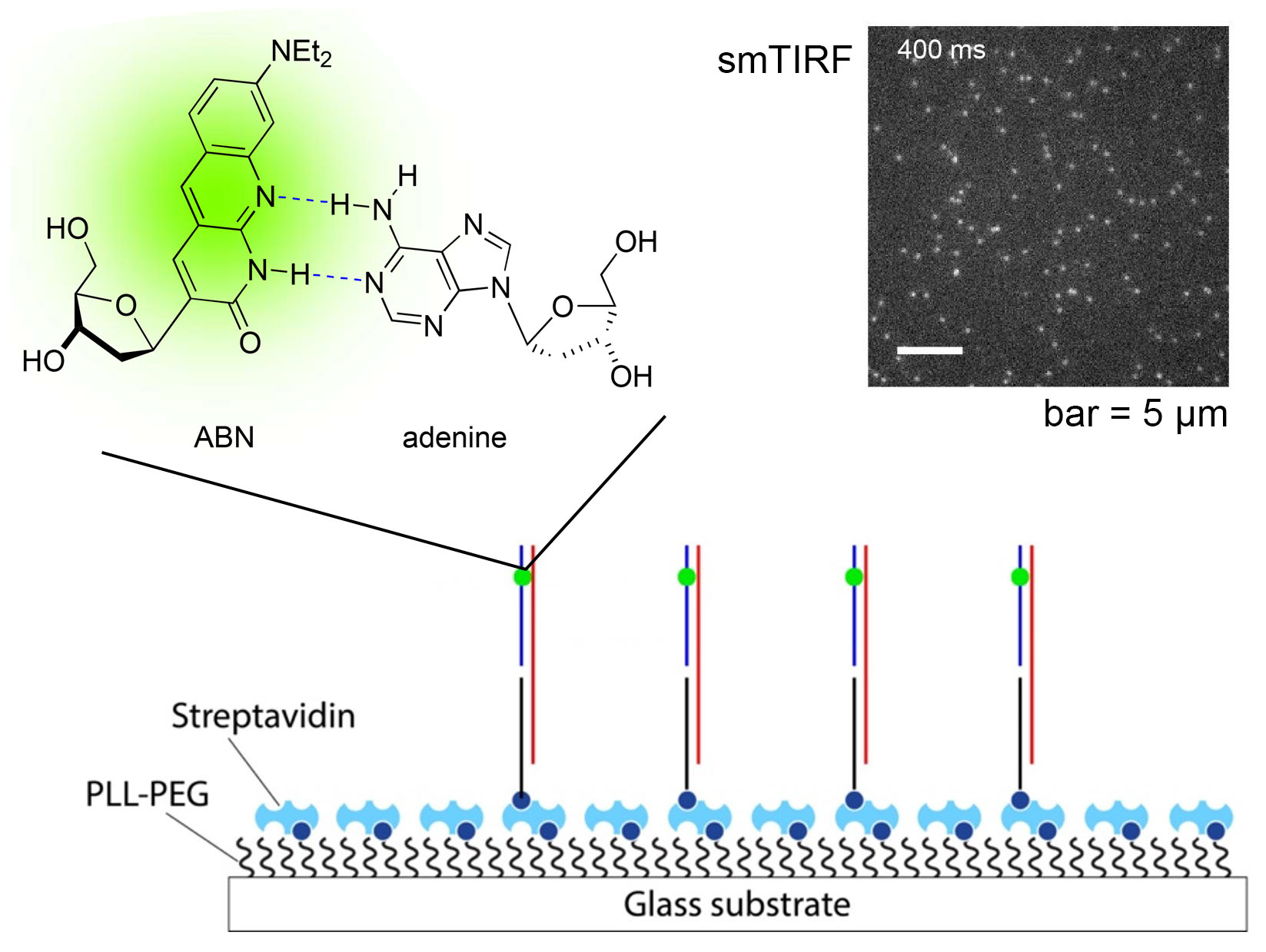
We’re thrilled to announce the publication of our latest paper, this one demonstrating that our fluorescent nucleobase analogue ABN provides very favorable spectral characteristics and quantum yield when based paired and stacked in duplex DNA oligonucleotides. By favoring a high quantum yield thymine-like tautomer through base pairing, surface-immobilized ABN-containing DNA duplexes are readily observed as bright spots using single-molecule fluorescence microscopy, exhibiting well-defined single-exponential bleaching kinetics. These results demonstrate that ABN is unique among FBAs in enabling single-molecule fluorescence studies of oligonucleotides using a standard microscopy setup.
Purse Lab paper: Bioconj. Chem. 2023, 34, 1601–1071.
Sequence-Specific Fluorescence Turn-On Sensing of RNA by DNA Probes Incorporating the Tricyclic Cytidine Analogue DEAtC
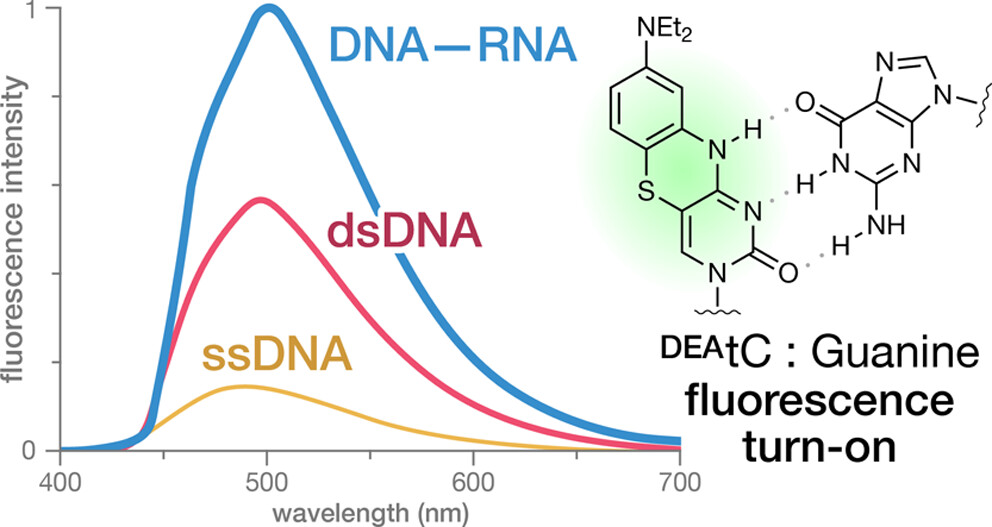
Purse Lab graduate students Ben Turner, Dana Rosansky, and Jesús Ceja, undergraduates Julian Cizmic and Grace Kim, and our collaborators in Tammy Dwyer’s lab at USD have published an exciting new study showing that synthetic DNA oligonucleotide probes containing our fluorescent nucleobase analogue DEAtC exhibit an average 8-fold increase in fluorescence intensity when hybridized to matched RNA with DEAtC base paired with G and little fluorescence turn-on when DEAtC is base paired with A. These sequence-specific fluorescence turn-on probes can immediately differentiate between match and mismatched sequences in hybrid duplexes.
Purse Lab paper: J Am Chem Soc 2022, 144, 14647–14656.
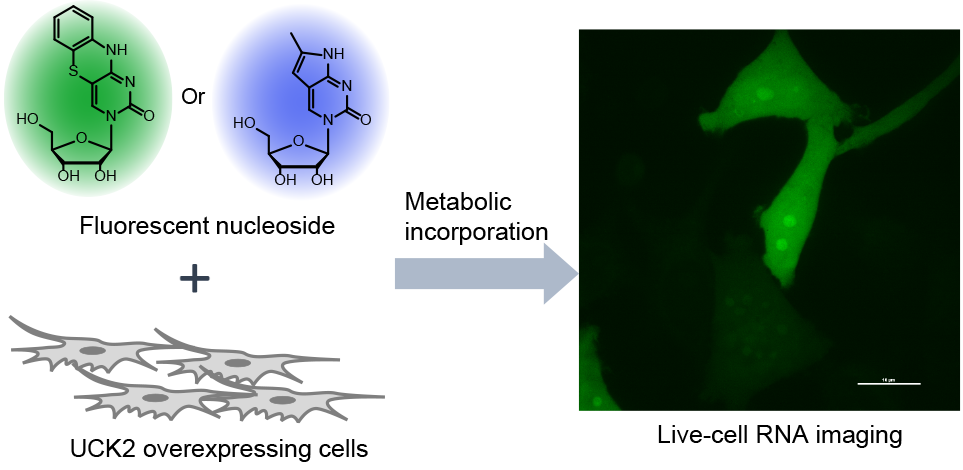
Ani Shalamberidze, a Ph.D. student member of our team, has published a JACS paper in collaboration with the Kleiner Lab at Princeton shows that a combination of judiciously selected fluorescent nucleoside analogues with genetic upregulation of nucleoside kinase activity enables metabolic labeling and live-cell imaging of RNA biology. The method has already been used to discover previously unknown cytoplasmic RNA granules generated during oxidative stress. Great ready for more discoveries in RNA biology!
Purse Lab paper: Chem Sci 2021, 12, 2623-2628.
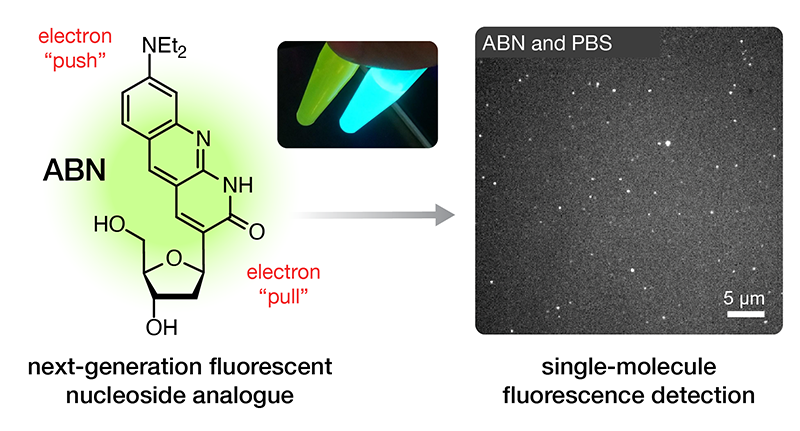
Purse Lab Ph.D. student George Samaan has develop the first fluorescent nucleobase analogue that performs robustly in single-molecule fluorescence measurements. Get ready for exciting new advances in single-molecule biophysical studies on nucleic acids!